Inside This Issue: HIV testing outcomes, U.S. HIV-2 diagnoses, long-acting HIV treatments, cure research, funding opportunities, global response to HIV, and educational resources.
The U.S. HIV Epidemic
HIV Testing Outcomes Among Blacks in Areas with High HIV Prevalence
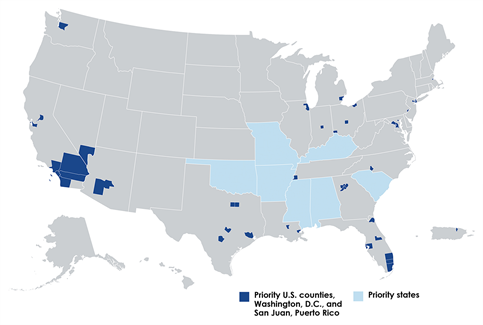
Blacks or African Americans (Blacks) have disproportionately high HIV infection rates. In 2017, 43% of new U.S. HIV diagnoses were among Blacks, who represent only 13% of the nation’s total population. For the U.S. as a whole, Blacks were also less likely than Whites to be linked to care within 90 days of their HIV diagnosis, retained in care, or be virally suppressed. The first phase of HHS’s new Ending the HIV Epidemic (EHE) initiative is targeting resources to 50 jurisdictions that accounted for more than half of all U.S. diagnoses and seven states with disproportionate HIV prevalence in rural areas (“Phase-1 jurisdictions,” shown in the map above). To gain a deeper understanding of the HIV epidemic among Blacks in these high prevalence areas, researchers from the Centers for Disease Control and Prevention (CDC) recently analyzed and summarized 2017 data on HIV testing, linkage to care, and partner services in Phase-1 jurisdictions.
The goal of U.S. HIV testing programs is to identify persons with HIV infection who are unaware of their status and to link all persons with HIV-positive test results to services. The CDC analysis showed that nearly two-thirds (62.9%) of CDC-funded HIV tests and 68.9% of new diagnoses were in Phase-1 jurisdictions. In these jurisdictions, Blacks accounted for about 40% of all HIV tests and nearly half (47.5%) of new diagnoses. Blacks also accounted for 58.5% of persons with a previous HIV diagnosis who were not known to be in care. Compared with Whites, a higher percentage of blacks in Phase-1 jurisdictions received a new diagnosis or had previously received a diagnosis and were not known to be in care.
Although 79.2% of Blacks with newly diagnosed HIV infection in Phase-1 jurisdictions were linked to HIV medical care within 90 days in 2017, this percentage was below the 2010 National HIV/AIDS Strategy (NHAS) goal of 85%. “This outcome suggests that the 2020 NHAS goals of 85% linkage within 30 days of diagnosis and the EHE initiative to reduce new HIV infections by 90% by 2030 might be challenging to achieve among Blacks without expanding current efforts and implementing novel testing strategies,” according to the study. “To achieve the goals of EHE, HIV prevention programs should focus on locally tailored evidence-based testing strategies to enhance and overcome barriers for linkage to and retention in care and reduce onward HIV transmission and HIV-related disparities.”
HIV-2 Infection
CDC Study Confirms that HIV-2 Diagnoses Remain Rare in the U.S.
Since 2014, CDC has recommended using an antibody-based HIV-1/HIV-2 differentiation test as part of a laboratory-based algorithm to confirm HIV-1 and HIV-2 infections. To examine trends both in HIV testing and in HIV-2 diagnoses, CDC researchers recently conducted a detailed analysis of the approximately 328,000 HIV diagnosed infections in the U.S. from 2010 through 2017, as well as the types of testing methods used in these diagnoses. Their analysis found that confirmed or probable HIV-2 diagnoses remain rare – totaling only 198 (0.06%) of all U.S. diagnoses during the period.
The researchers also found that, although the use of the HIV-1/HIV-2 differentiation test increased steadily throughout the study period, the number of confirmed or probable HIV-2 infections remained stable after 2014. In addition, the number of persons with confirmed HIV-1 infection who had a false-positive HIV-2 test result on the HIV-1/HIV-2 differentiation test was greater than the total number of confirmed and probable HIV-2 diagnoses combined. The researchers believe that HIV antibody cross-reactivity was the likely cause of the false-positive reactions, which required additional time and testing to resolve.
Based on this analysis, the CDC researchers conclude that “updates to the laboratory-based [HIV] testing algorithm merit consideration in the United States. This could include development of new FDA-approved tests to reduce the time to HIV diagnosis and treatment, primarily for HIV-1, but in rare cases, for HIV-2.”
%20small.jpg)
New and Experimental HIV Treatments
NIAID Clinical Trial Evaluates Antibody-Drug Combination for Long-Acting HIV Treatment
The National Institute of Allergy and Infectious Disease (NIAID) recently launched the first clinical trial to test the combination of an anti-HIV drug plus an HIV antibody as a long-acting treatment for people living with HIV. In the NIAID-funded Phase 2 trial called A5357, researchers are studying the safety and efficacy of the experimental integrase inhibitor cabotegravir plus a powerful genetically engineered antibody called VRC07-523LS. The current treatment for HIV typically involves taking two or more antiretroviral drugs (ARVs) in tablet form daily, often as a single multidrug pill. Since some people have difficulty adhering to a daily ARV regimen, researchers have been studying new long-acting HIV regimens that would only require monthly – or even less frequent – doses.
Two Phase 3 clinical trials have already shown that a monthly injection of long-acting formulations of the ARVs cabotegravir and rilpivirine can effectively treat HIV in adults. Instead of combining two ARVs, the new A5357 trial combines long-acting cabotegravir with broadly acting neutralizing antibodies (bNAbs) that have been shown in lab studies to stop a wide variety of HIV strains from infecting immune cells.
Participants in the A5357 study will discontinue part of their current ARV regimen and start taking daily oral cabotegravir. Those who tolerate cabotegravir and still have a suppressed HIV viral load about a month after starting the trial will discontinue daily oral ARVs and start receiving the experimental treatment regimen. This will consist of an injection of long-acting cabotegravir once every 4 weeks and an intravenous infusion of VRC07-523LS once every 8 weeks. Participants will remain on this experimental treatment regimen for 44 weeks. The study team will measure the participants’ HIV viral load every 2 to 4 weeks. At the end of this 44-week study period – or sooner if the viral load rebounds to a detectable level – participants will resume taking daily oral ARV therapy and will have their viral load monitored for an additional 48 weeks. The results of the study are expected by 2023.
Cure Research
Researchers Reverse HIV and SIV Latency in Two Animal Models
The National Institutes of Health (NIH) reports that, in a range of experiments, scientists have successfully reactivated resting immune cells in the bloodstream or tissues of animals that were latently infected with HIV or a related virus called SIV that infects monkeys. These reactivated immune cells started making copies of the viruses, which could potentially be neutralized by anti-HIV drugs and the immune system. According to NIH, this advance “marks progress toward a widely accessible cure for HIV.”
Although current antiretroviral treatment (ART) regimens maintain the health of people with HIV and prevent transmission of the virus, they are not a cure. Developing an HIV cure has been very difficult due to the persistence of “viral reservoirs” – places where HIV may remain hidden from the immune system. “These reservoirs consist of HIV-infected cells containing HIV genetic material that can generate new virus particles if a person’s treatment is interrupted,” according to NIH. “The cells have entered a resting state that they maintain until they are activated to produce the virus. The immune system cannot recognize and kill HIV-infected cells in a resting state, and ART has no effect on them.”
To overcome this challenge, researchers have been pursuing a strategy called “kick and kill” which involves activating HIV viral reservoirs so that drug therapies or an enhanced immune system can recognize and kill HIV-infected cells, thereby eliminating HIV. Unfortunately, previous attempts to reactivate or “kick” the HIV reservoir worked well in the laboratory but were either ineffective or too toxic when tested in animals and people.
“A simple, safe, and scalable cure for HIV is an aspirational goal that, if achieved, would accelerate progress toward ending the HIV pandemic,” notes Dr. Anthony Fauci, director of the NIAID. “These new findings help sustain our cautious optimism that an HIV cure is possible.”

Ending the HIV Epidemic
CDC Announces Major HIV Funding Opportunity Focusing on Heavily Affected Communities
Late last month, CDC announced a five-year funding program titled Integrated HIV Programs for Health Departments to Support Ending the HIV Epidemic in the United States (PS20-2010). “This funding opportunity can help end this epidemic by infusing communities with the resources, technology, and expertise needed to strengthen HIV prevention and treatment,” according to CDC. “To achieve maximum impact, the funding targets geographic areas that account for more than half of new HIV diagnoses, and states with a substantial rural burden. In recent months, each of these jurisdictions has developed a comprehensive Ending the HIV Epidemic plan, informed by consultations with diverse stakeholders and community-level input.” First-year awards under this funding opportunity are expected to total about $109 million. Additional details about this funding opportunity are available on the CDC website. The closing date for applications is March 25, and the anticipated award will begin on June 1.

Global HIV Response
New Business Alliance Will Support Efforts to End AIDS by 2030
In late January, UNAIDS announced the launch of the Business Alliance to End AIDS by 2030 (Alliance) , a public-private coalition “to strengthen cross-sector collaboration, build on common values, and create spaces for effective collaboration,” according to UNAIDS. The Alliance “aims to provide businesses with the necessary tools and support needed from public sector partners to help realize the goal of ending AIDS by 2030.” In addition, the Alliance plans to help shape regulations and policies that support HIV prevention and treatment programs and that end HIV-related stigma and discrimination.
“Today, every single company must make a positive contribution to the societies they serve – this is not an option, this is an imperative,” notes Winnie Byanyima, executive director of UNAIDS. “Beyond reaching their economic objectives, companies must deliver social and environmental impact. The private sector has a key role to play in the multisectoral response to HIV. Businesses must ensure that their employees, their suppliers, and communities receive life-saving HIV treatment, prevention, and care programs. Together, we can end AIDS by 2030.”

Educational Resources
TAG Compiles Resources on HIV and Aging & Cure Research
The Treatment Action Group (TAG) recently updated its extensive online resources focusing on HIV and aging and HIV cure research:
HIV and aging: This resource listing includes links to aging-related trials and research studies; selected scientific publications; community-based articles and reports; mainstream media articles; general resources and guidelines; and news from conferences and meetings.
Cure-related research resources: This resource is similar in scope to the HIV and aging resource above. It includes many articles, reports, websites, and ongoing clinical trials related to the research effort to cure HIV infection.
Research toward a cure trials: This resource links to clinical trials and observational studies related to the research effort to cure HIV infection, mainly derived from the clinicaltrials.gov online registry. By clicking on the trial registry identifier numbers for specific trials, users can access detailed information on the study design, enrollment criteria, principal investigators, and location(s).
If you have suggestions for additional links for these pages, please email Richard Jefferys, TAG’s director of basic science, vaccines, and cure research: richard.jefferys@treatmentactiongroup.org
AIDSinfo Updates Fact Sheets on Investigational HIV Drugs & Living with HIV
HHS has recently posted several updated fact sheets about investigational drugs and living with HIV on its AIDSinfo site. The investigational drug fact sheets are available in two formats: a plain-language version suitable for patients, and a more technical version for health professionals. Each plain-language version includes an overview of the investigational drug and information about clinical trials studying the drug. The health professional version provides more detailed scientific information about the drug, including its pharmacology and side effects seen in clinical trials. The list of fact sheets below provides each drug name and a brief description, together with links to the patient and health professional versions of each fact sheet:
- Lexgenleucel-T (gene therapy product): patient version, health professional version
- Panobinostat (latency-reversing agent): patient version, health professional version
- Romidepsin (latency-reversing agent) : patient version, health professional version
AIDSinfo’s updated consumer fact sheets, which are available in English and Spanish, provide important information for people living with HIV. Each fact sheet has a summary of key points together with links to additional information and resources.



